We also identify the gaps in our current understanding and highlight important topics for future research. 
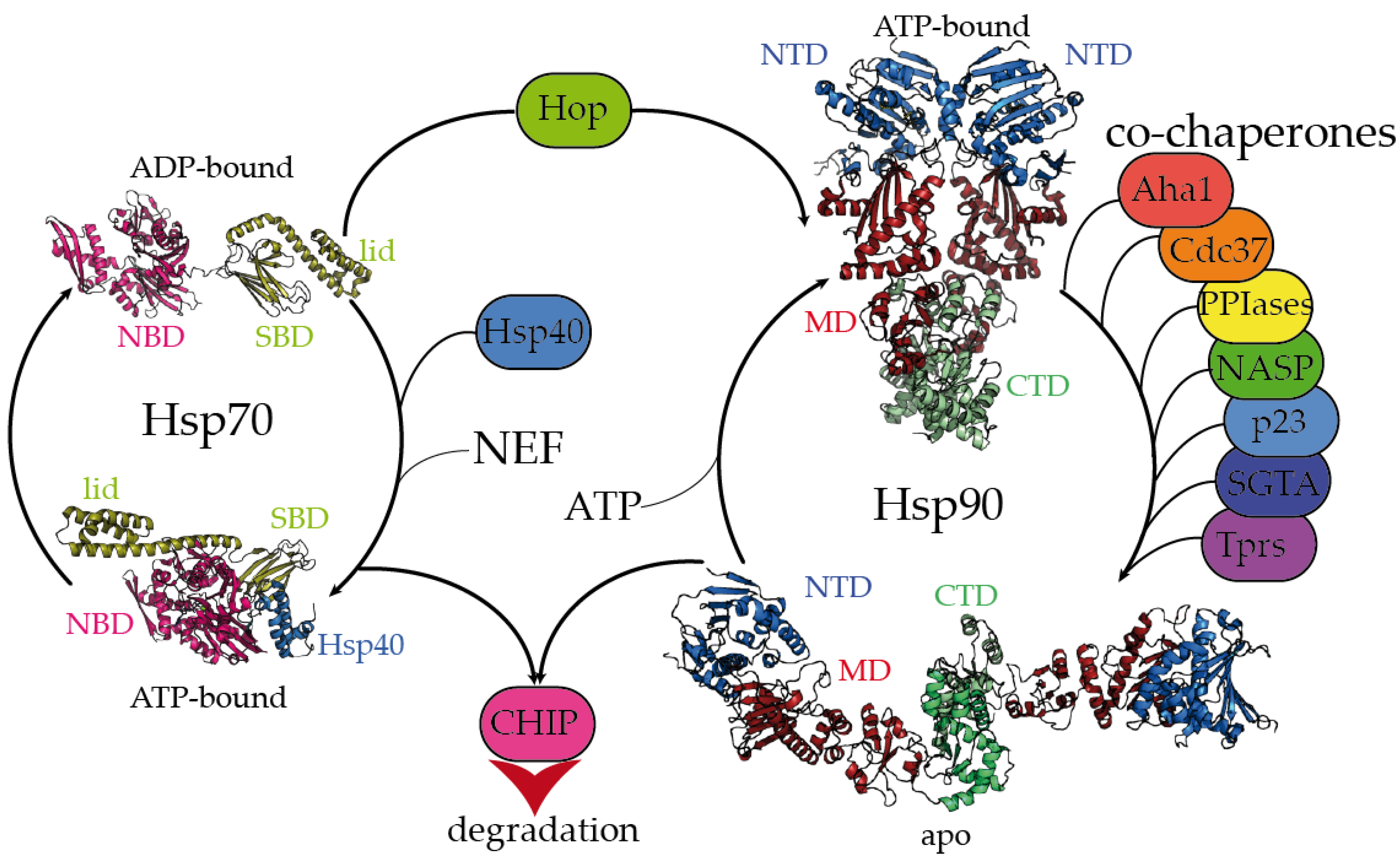
In addition we describe the exciting progress that has been made in the development of Hsp90 inhibitors, which are now showing promise in the clinic for cancer treatment. Here we review the biology of the Hsp90 molecular chaperone, emphasizing recent progress in our understanding of structure–function relationships and the identification of new client proteins. Targeting the Molecular Chaperone Hsp90 in Cancer: What Does the Biology Tell Us TAT 2011. Here, we demonstrate that HSP90 plays a powerful oncogenic role by inducing p14ARF degradation, which prevents cellular senescence. The cycle is also regulated by a group of co-chaperones and accessory proteins. 
Hsp90 operates as a dimer in a conformational cycle driven by ATP binding and hydrolysis at the N-terminus. Treating KRAS(G12D) inhibitor resistance using a KRAS- and HSP90 chaperone-targeted hetero-bispecific small molecule agent abstract. By this means, Hsp90 displays a multifaceted ability to influence signal transduction, chromatin remodelling and epigenetic regulation, development and morphological evolution. It interacts with ‘client proteins’, including protein kinases, transcription factors and others, and either facilitates their stabilization and activation or directs them for proteasomal degradation. Beyond these specific regulators, the Hsp90 molecular chaperone, a member of the general cellular protein-folding machinery, also plays a fundamental role in the regulation of many kinases. The molecular chaperone Hsp90 (90 kDa heat-shock protein) is a remarkably versatile protein involved in the stress response and in normal homoeostatic control mechanisms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
